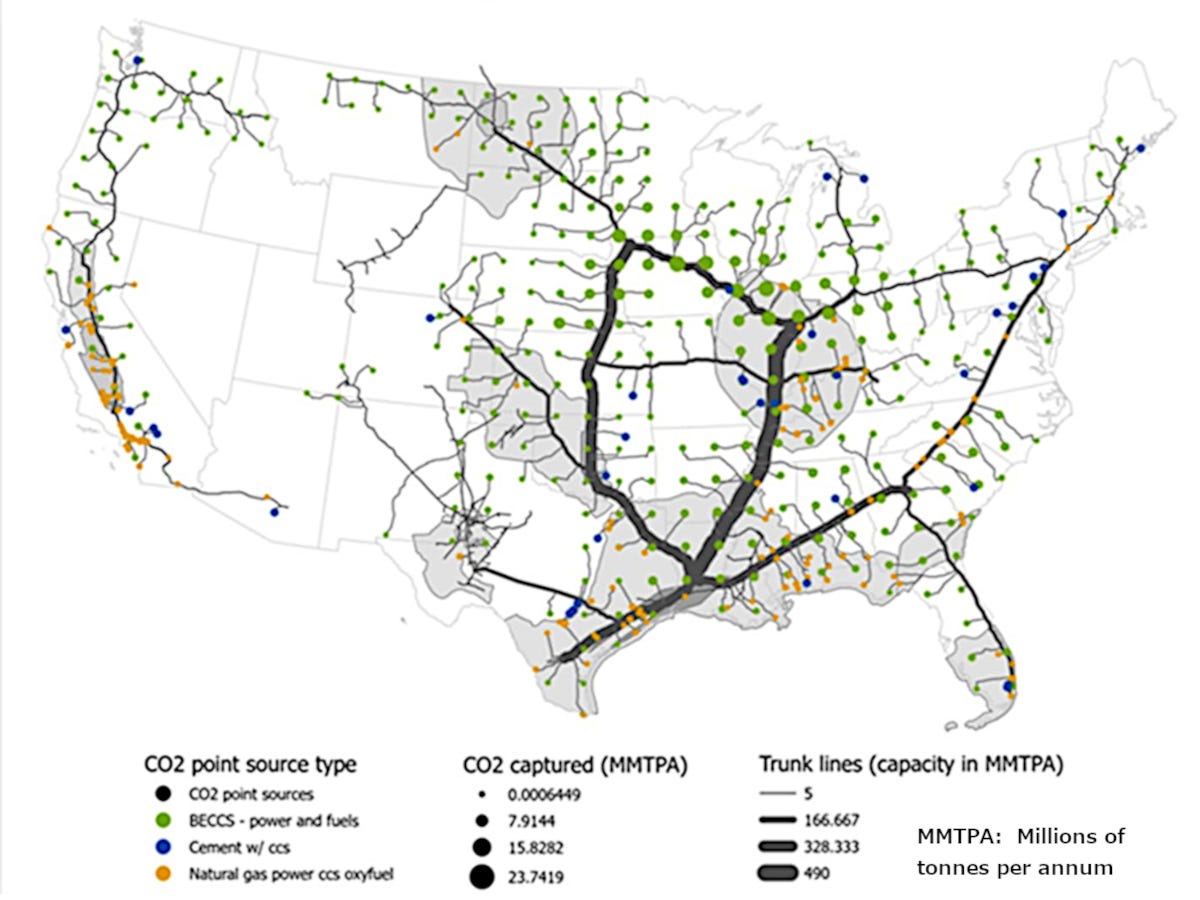
Carbon Dioxide Pipelines
What could possibly go wrong?
Previously, it was speculated that the experience of the fossil fuel industry might cause us to consider limits on the rate at which we inject CO2 into the ground, concluding that if the behavior of CO2 could be modeled as equivalent to waste water, we would need some 3725 dispersed CO2 sequestration wells for each gigaton per year of CO2 to be sequestered, to potentially avoid causing earthquakes. Per the EPA [1]the US power plants alone produced 1.7 gigatons of CO2 in 2022, and 1 part per million CO2 in the atmosphere equates to about 7.8 gigatons. So it is going to take many more pipelines than shown above, if the US is going to contribute its fair share to CO2 reduction via carbon capture and sequestration (CCS) [2].
The need for CO2 pipelines for CCS brings with it a number of problems, including the disproportionate impact on poor and indigenous communities that has typified the history of pipeline construction [3]. Some of that might be mitigated by repurposing existing natural gas pipelines, but the known technical problems, though not trivial, are inconsequential in comparison with the problem of repurposing itself, which inherently entails curtailing the use of natural gas as a fuel. That is to say, it is primarily a political problem.
Although CO2 is not explosive and not poisonous, per se, CO2 pipelines pose dangers which are in some respects greater than those conveying "natural" gas, which is over 85% methane. A bit of chemistry and physics is required to explain why.
The molecular weights of methane, air and carbon dioxide are approximately 16, 29, and 44, respectively. Thus methane is lighter than air, while CO2 is heavier. Leaking CO2 will tend to pool near the ground as it diffuses while natural gas will rise. CO2 is very soluble in water, with a Henry constant [4] of 334E-4 mol/l atm at 25C (compared to values of 13.2E-4 for O2 and 14.2E-4 for methane). Also: the effects of wind diminish as one gets closer to the surface of the earth. This so-called boundary layer effect is predicted by stipulating continuity of the velocity field, which necessitates a velocity of zero at any fixed surface. With these few facts, plus Dalton's law (The partial pressure of a gas is proportional to its molar concentration) and Henry's law (The equilibrium concentration of a dissolved gas is proportional to its partial pressure above the liquid) we are ready to explore the consequences of a CO2 pipeline burst or major leak under a body of water, and compare that with a similar natural gas event.
If the leak involves a natural gas pipeline, the gas transits the water and disperses above, unless of course there is something around to ignite it. But, neglecting impurities, there is no effect on the water, and people nearby are unlikely to be harmed, unless there is a fire or explosion. This is the result of two factors: methane's being lighter than air, so no boundary layer is formed, and its very low atmospheric concentration, leading to a correspondingly low dissolution in water. In the case of CO2, however, the situation is altogether different. As the CO2 reaches the surface, a boundary layer of higher CO2 concentration is formed - even if a wind is blowing. This triggers a rapid and large increase in the CO2 concentration in the water, and simultaneously the outgassing of O2 from the water. The EPA has approved use of CO2 as a pesticide to deal with certain species of invasive fish [5] but their internal studies showed toxicity effects on other than the target species. They offered no explanation as to why some species seem to be substantially less subject to the toxic effects of dissolved CO2 than others, but there is an easy one. Many varieties of fish are able to oxygenate the water surrounding their gills by going to the surface and gulping air. This allows them to survive even in the absence of dissolved oxygen. However, in our accident scenario, this does not help, because the surface layer becomes very poor in oxygen. So, in other words, when there is an underwater CO2 pipeline burst or major leak, all the aquatic animals in the vicinity are likely to die.
Then there are the effects of the CO2 on animals on land. Here, the simple calculations behind the foregoing discussion are inadequate; it is necessary to use computational fluid dynamics (CFD) A study in 2018 [6] involved CFD modeling in conjunction with an experiment in which a pipe containing pressurized CO2 was explosively ruptured. The model predictions and test results were in reasonable agreement, and indicated that even in the presence of a 7 mph wind CO2 concentrations exceeded the OSHA threshold [7] for dangerous exposure for a minute or more at a distance of 100 meters downwind. The reality of the risk was demonstrated in 2020 near the village of Satartia MS [8] when the high airborne CO2 concentrations following a CO2 pipeline burst sent 45 people to the hospital.
Perhaps as a result of that accident, in June 2022 the Biden administration announced plans [9] to address the risks of CO2 pipelines [10]. Yet there is pressure [11] to "streamline" the CO2 pipeline and well approval process. Experience with past pipeline projects being railroaded despite violations of treaties and disregard of environmental consequences together with the novel risks associated with CO2 pipeline accidents suggest that we should do the opposite: Take deliberate care that the routing and technical designs of all CO2 pipelines account not only for physical safety but also the rights and interests of the populations affected. Priority must be given to repurposing existing natural gas pipelines, both to speed the transition away from fossil fuels and to avoid the needless damage to nature, together with increased CO2 emissions due to indirect land use change, that occurs when a new pipeline right of way is cleared. Finally, there should be a requirement that new large point source facilities and direct air capture plants be located in close proximity to sequestration sites, to minimize pipeline footprints.
Notes
[1] https://www.epa.gov/power-sector/power-plant-emission-trends
[2] As an aside, I do not advocate solving the CO2 problem solely with CCS. But there is a role for it.
[3] For instance, the DOE sponsored a workshop on Carbon transport and storage in February 2022 to address R&D priorities for repurposing the infrastructure https://www.energy.gov/sites/default/files/2022-11/%5BWORKSHOP%5D-Carbon-Transport-and-Storage-R%26D-Priorities-for-Repurposing-Infrastructure.pdf, inviting over 170 government and industry stakeholders but apparently no civic, environmental or indigenous peoples representatives.
[4] https://acp.copernicus.org/articles/15/4399/2015/
[6] https://www.sciencedirect.com/science/article/pii/S1876610219307246
[7] 4% concentration is the OSHA threshold for dangerous CO2 exposure; at 10% concentration CO2 is an asphyxiant.
[8] https://www.npr.org/2023/05/21/1172679786/carbon-capture-carbon-dioxide-pipeline
[9] https://www.eenews.net/articles/biden-releases-plan-to-avoid-dangerous-co2-pipeline-failures/
[10] One of the ways suggested by the DOE to reduce risk is to dope the CO2 with an odorant such as H2S. Yet a CFD study https://ro.uow.edu.au/cgi/viewcontent?article=1997&context=eispapers1 of 95% CO2/5% H2S plumes indicated larger zones of fatality due to H2S poisoning than to CO2 asphyxiation.
[11] https://clearpath.org/our-take/need-for-speed-removing-roadblocks-for-co2-pipelines-and-wells/
Reposted 19 February 2024 with formatting changes